Spinal cord stimulator (SCS)
 A spinal cord stimulator (SCS), also known as a dorsal column stimulator, is a device surgically placed under your skin to send a mild electric current to your spinal cord. This therapy can essentially reduce your pain significantly.
A spinal cord stimulator (SCS), also known as a dorsal column stimulator, is a device surgically placed under your skin to send a mild electric current to your spinal cord. This therapy can essentially reduce your pain significantly.
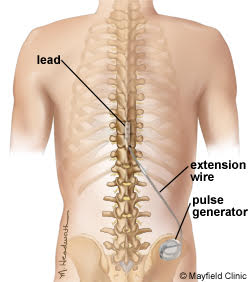
Stimulation does not eliminate the source of pain, it simply interferes with the signal to the brain, therefore, the amount of pain relief varies for each person. The device is first done as a trial before it is placed permanently. The goal for spinal cord stimulation is a 50-70% reduction in pain. However, even a small amount of pain reduction can be significant if it provides you with quality of life and a decrease or elimination of your pain medications.
If the trial is unsuccessful, the implant can be removed and does not damage the spinal cord or change the anatomy.
FDA-approved indications include the following:
1) Failed Back Surgery Syndrome (FBSS)
2) Chronic painful peripheral neuropathy
3) Multiple Sclerosis (MS)
4) Complex Regional Pain Syndrome (CRPS) I and II
You may be a candidate for SCS if you meet the following criteria:
*This procedure is performed by Dr. Bannister as an outpatient procedure and is covered by insurance.

